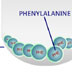
Animation 22: DNA words are three letters long.
Marshall Nirenberg and Har Khorana crack the genetic code.
I'm Marshall Nirenberg. Har Khorana's group and mine "cracked" the genetic code. We figured out how the nucleotide language of mRNA is "translated" into the amino acid language of proteins. Genetic data from Crick and others showed that three nucleotides form a "codon" — an mRNA word that specifies one amino acid. This made sense, because a codon made from only one or two nucleotides would not produce enough combinations (words) to code for all 20 of the known amino acids. However, a three-nucleotide codon produces 64 combinations. This would produce a redundant, or degenerate, code where several different codons specify the same amino acid. The parsimony principle — that the simplest solution is often right — ruled out a four-nucleotide codon. In 1961, Heinrich Matthaei, a visiting post-doc, and I started experiments to test the triplet codon hypothesis. We used a cell-free extract of E. coli because we believed that this extract should contain all the components needed to translate mRNA into proteins. The extract was treated with DNase to destroy any remaining E. coli DNA — so there was no template from which messenger RNA could be made. Using a method perfected by Marianne Grunberg-Manago and Severo Ochoa, we made synthetic mRNA composed entirely of uracil. We added this poly-U RNA to the extract. When we examined the products produced in this cell-free system, we found polypeptides composed entirely of the amino acid phenylalanine (PHE). From this result, we concluded that a sequence of three uracils — UUU — must code for phenylalanine. We had determined one of the 64 triplet codons! Matthaei and I tried other polynucleotide chains. Poly-C made a proline (PRO) chain; poly-A made a lysine (LYS) chain. Interestingly, no protein was made with the poly-G chain. Other researchers and I quickly recognized the power of this approach. Using RNA templates containing different nucleotide combinations, we assigned amino acids to about 50 triplet codons. This is a genetic code table. To find a specific codon, look down the left hand side for the first letter of the codon. For the second letter, look across the table. For the last letter, look down the right hand side of the table. For example, the codon C G A: The codon C G A codes for the amino acid arginine (ARG). Hi, I'm Phil Leder. I helped Marshall Nirenberg with the rest of the genetic code. Some of the codons were hard to decipher, because we couldn't establish their order biochemically. For example, a triplet with two Gs and one C could be in the order CGG or GCG or GGC. Marshall and I used the activation of tRNA to solve this problem. tRNA is the molecule that carries amino acids to the ribosomes for protein synthesis. In 1962, Robert Holley solved the structure of tRNA. Although tRNA is single stranded, stretches of complementary nucleotides hydrogen bond to form short double-stranded regions, which bend the tRNA into a characteristic cloverleaf shape. Holley showed that all tRNAs have a similar cloverleaf structure. At a position on one of the "leaves," a sequence of three nucleotides forms an anti-codon, which base pairs with a specific mRNA codon. Thus, there is a different tRNA molecule corresponding to each mRNA codon. Using their cell-free translation system, Zamecnik and his group showed that tRNA becomes activated when the amino acid binds to the tRNA's stem. This step requires chemical energy in the form of ATP. I first made short RNA chains composed of three or six nucleotides. When added to a cell-free extract, an activated tRNA will read these trinucleotide or hexanucleotide sequences. After that, all I had to do was make specific trinucleotide and hexanucleotide chains to confirm the genetic code. We also found that translation starts with a specific codon AUG — the only unique codon — and there are stop codons that end translation. So, in addition to words, the genetic code also has punctuation points. So, to put this all together: within the nucleus, the DNA code is transcribed into a complementary mRNA molecule. The mRNA enters the cytoplasm, where it associates with a ribosome. The mRNA code is then translated into a polypeptide chain. The codon AUG signals the start of translation. An activated tRNA ferries the first amino acid — methionine — to the ribosome. The tRNA anti-codon binds to the AUG codon on the mRNA. The whole complex shifts, and the next codon is read by another tRNA. As the two amino acids are held in position, a peptide bond is formed between them. The second tRNA accepts the growing protein chain, and the methionine tRNA is released. The process is repeated until a stop codon is encountered. When the stop codon, in this case UAA, is reached, translation is finished. Stop codons do not have matching tRNAs. The ribosome disassembles to be reused for translating another mRNA and one complete peptide chain is released.
marshall nirenberg, severo ochoa, rna templates, messenger rna, amino acids, codon, genetic code
- ID: 16494
- Source: DNALC.DNAFTB
Related Content
15353. Figuring out the other codons, Marshall Nirenberg
After decoding the "easy" codons, Marshall Nirenberg talks about his strategy for decoding the rest.
15350. The RNA code for phenylalanine, Marshall Nirenberg
Marshall Nirenberg talks about the RNA code for phenylalanine.
16511. Biography 22: Marshall Warren Nirenberg (1927- )
Marshall Nirenberg, Har Gobind Khorana, and Robert Holley shared the 1968 Nobel Prize for Physiology and Medicine. Nirenberg and Khorana cracked the genetic code. Holley sequenced and deduced the structure of the first tRNA molecule.
15349. RNA can stimulate protein synthesis, Marshall Nirenberg
Marshall Nirenberg talks about RNA can stimulate protein synthesis.
15356. Determining the nucleotide sequences of all RNA codons, Marshall Nirenberg
Marshall Nirenberg talks about Gobind Khorana, who synthesized many of the triplets needed to finish the decoding process.
15882. Breaking the code
Marshall Nirenberg and Heinrich Matthaei used poly-U mRNA in a cell-free system to make a polyphenylalanine protein chain. This showed that UUU must be the code that specifies the amino acid phenylalanine.
15354. Origin of the RNA code, Marshall Nirenberg
Marshall Nirenberg talks about origin of the RNA code.
15481. Translation: RNA to protein, 3D animation with no audio
Translation: RNA to protein, 3D animation with no audio
15885. Cell-free extracts
Paul Zamecnik first developed the cell-free extract system, which Marshall Nirenberg adapted to decipher the genetic code. Paul Zamecnik and Mahlon Hoagland also isolated activated tRNA, the "adaptor" that shuttled amino acids to ribsomes for incorporati
15501. Translation: RNA to protein, 3D animation with basic narration
3D animation of translation: RNA to protein.